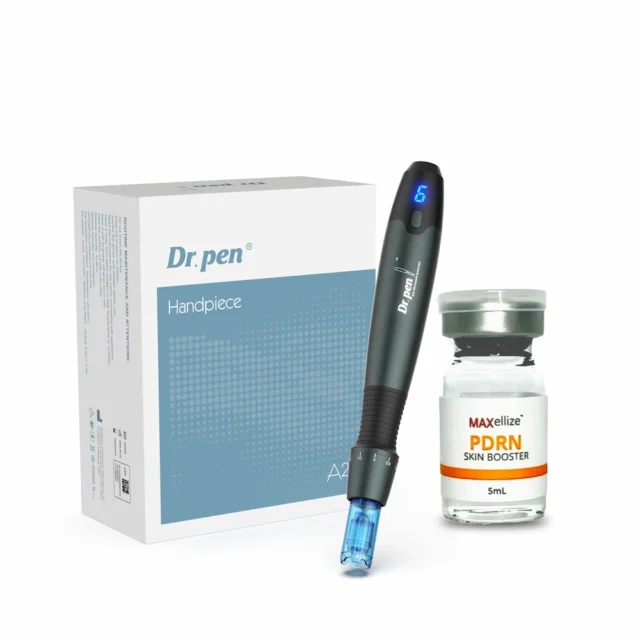
Dr. Pen A20 – FDA CLEARED – professional grade
Dr. Pen A20 Technical Specifications
Face Med Store provides the complete A20 system with documented specifications validated through FDA 510(k) clearance (K230420).
| Specification | Value |
|---|---|
| Model | A20 |
| FDA Classification | Class II Medical Device |
| Product Code | QAI |
| Regulation Number | 21 CFR 878.4430 |
| Motor Speed | 6300-7700 RPM |
| Stamp Rate | 105-128 stamps/second |
| Needle Count | 14 per cartridge |
| Needle Gauge | 34 Ga surgical-grade stainless steel |
| Needle Spacing | 2mm radial arrangement |
| Depth Settings | 9 levels (0mm to 2.0mm in 0.25mm increments) |
| Recommended Treatment Depth | 1.5mm |
| Power Source | Rechargeable Li-ion battery |
| Cartridge Shelf Life | 2 years |
How the Dr. Pen A20 Stimulates Collagen Production
Face Med Store’s A20 system triggers percutaneous collagen induction, a controlled wound-healing response that rebuilds damaged dermal architecture.
The 34-gauge needles penetrate the stratum corneum at your selected depth setting. This penetration creates thousands of micro-channels per square centimeter.
Micro-channels activate platelet degranulation, which releases growth factors including TGF-β and PDGF. Growth factors recruit fibroblasts to the treatment zone. Fibroblasts synthesize new collagen and elastin fibers over the following 4-12 weeks.
The Dr. Pen A20’s 2mm needle spacing ensures optimal coverage without overlapping trauma zones, which means your patients experience faster healing with less post-treatment erythema.
Dr. Pen A20 Needle Cartridge System
Face Med Store includes 10 sterile needle cartridges with every A20 kit. Each cartridge features ethylene oxide sterilization and single-use design to eliminate cross-contamination.
| Cartridge Feature | Clinical Benefit |
|---|---|
| 14 radially-arranged needles | Consistent micro-channel distribution |
| 34Ga surgical-grade steel | Minimal epidermal tearing |
| 2mm inter-needle spacing | Reduced treatment overlap |
| EtO sterilization | Verified sterility assurance |
| Secure bayonet attachment | No mid-treatment dislodging |
| Disposable sleeve system | Prevents device contamination |
The A20 cartridges maintain a 2-year shelf life when stored at room temperature.
Dr. Pen A20 Treatment Protocol
Face Med Store recommends following this evidence-based protocol for facial acne scar treatment:
- Cleanse the treatment area with antiseptic solution and apply topical anesthetic for 20-30 minutes
- Select the appropriate depth setting based on skin thickness and scar severity (typically 1.0-1.5mm for facial acne scarring)
- Attach a new sterile cartridge and confirm secure connection
- Apply the disposable protective sleeve to the device body
- Hold the A20 perpendicular to the skin surface and move in vertical, horizontal, and diagonal passes
- Monitor for pinpoint bleeding, which indicates adequate penetration depth
- Apply post-procedure serum containing hyaluronic acid or growth factors to maximize transdermal delivery
- Dispose of the cartridge and sleeve in a sharps container
The Dr. Pen A20 requires 4-6 treatments spaced 4-6 weeks apart for optimal acne scar revision results.
Why Practitioners Choose the Dr. Pen A20
Face Med Store supplies the A20 to aesthetic practices seeking FDA-cleared collagen induction therapy with documented clinical validation.
Your patients want visible improvement. The A20 delivers 6300-7700 RPM precision that manual rollers cannot match.
You need regulatory compliance. The A20 carries 510(k) clearance under 21 CFR 878.4430.
You deserve reliable equipment. The A20’s rechargeable Li-ion battery provides consistent power throughout multi-area treatments.
Face Med Store’s A20 system transforms acne scar treatment from guesswork into measurable outcomes.
Dr. Pen A20 Complete Kit Contents
Face Med Store ships every A20 with these components:
| Component | Quantity |
|---|---|
| Dr. Pen A20 device body | 1 |
| Li-ion battery charger/adapter | 1 |
| Sterile needle cartridges | 10 |
| Protective cartridge covers | 10 |
| Disposable device sleeves | 10 |
| User documentation | 1 set |
Dr. Pen A20 Safety and Regulatory Compliance
Face Med Store only distributes devices meeting rigorous quality standards.
The Dr. Pen A20 has achieved:
– FDA 510(k) clearance (K230420)
– Electrical safety certification per IEC 60601-1
– Electromagnetic compatibility verification
– Biocompatibility testing per ISO 10993
– Performance validation per FDA guidance
The A20’s single-use cartridge design eliminates needle reuse, which means zero patient-to-patient contamination risk.
## Order the Dr. Pen A20 from Face Med Store
Face Med Store offers the Dr. Pen A20 exclusively to licensed healthcare practitioners and aesthetic professionals.
Stop compromising with inconsistent microneedling devices. Start delivering FDA-cleared collagen induction therapy your patients can trust.
**Price available to professional users only**
Disclaimer
No warranties or representations. Sellers and purchaser specifically acknowledge that except as otherwise set forth in this purchase agreement, sellers are selling and purchaser is purchasing the purchased equipment/tools “as is, where is.” Seller is not providing any warranty for the product and not making any warranty, express or implied. Purchaser has inspected the equipment/tool, and is not relying on any representations or warranties of any kind.
Purchaser agrees and understands that it is solely the responsibility of the purchaser to know and understand the training and regulatory requirements of the state in which she/he is operating in order to use the product. Purchaser agrees to defend and indemnify seller for any misuse of the product and seller shall in no way be responsible for any injury, accident, malfeasance or misfeasance related to purchaser’s use of the product.
Disclaimer of warranty
Purchaser agrees and understands that it is solely the responsibility of the purchaser to know and understand the training and regulatory requirements of the state in which she/he is operating in order to use the product. Purchaser agrees to defend and indemnify seller for any misuse of the product and seller shall in no way be responsible for any injury, accident, malfeasance or misfeasance related to purchasers use of the product.

Dr. Pen A20 – FDA CLEARED – professional grade
Price available to professional users only