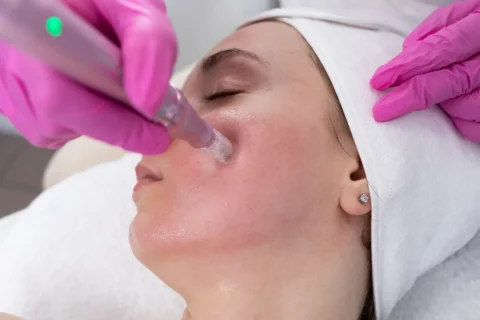
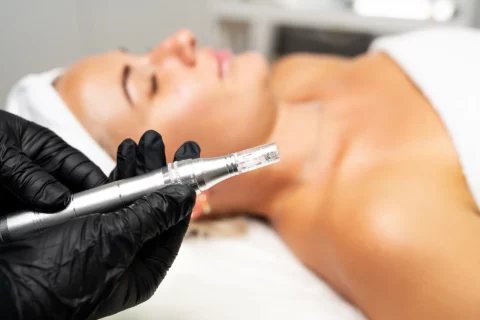
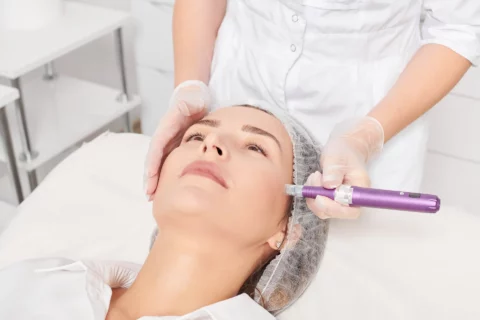
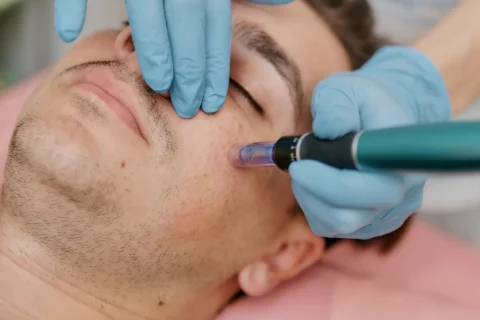
Microneedling Innovations: How Microneedling Pen Stands Out in the Industry
Microneedling Pen is an innovative microneedling device that has captured the interest of skincare and aesthetics enthusiasts worldwide. With its growing popularity and extensive use, one might wonder whether the device has received FDA approval.
In this article, we will explore the current FDA approval status of Microneedling Pen, discuss any safety concerns, and provide an overview of the benefits of using this cutting-edge device.
Get Everything You Need for Your Microneedling Pen and More at 20% off Your First Order with Code “20OFF”!
The Microneedling Pen treatment requires only the best equipment to deliver treatments and results your patients will love. Get your best Microneedling Pen equipment and more at FACE Med Store today!
You can create an account here.
Understanding the FDA’s Classification of Microneedling Devices: Safety and Effectiveness in Focus
While microneedling devices haven’t received blanket FDA approval for general use, the FDA has taken steps to ensure their safety and effectiveness in aesthetic applications. Specifically, the FDA classifies microneedling devices for aesthetic use as Class II devices, which are subject to special controls designed to provide a reasonable level of assurance regarding their safety and efficacy.
These special controls are outlined in the FDA’s final order and form an integral part of the regulatory framework for microneedling devices used for aesthetic purposes. It’s important to note that this guidance does not extend to dermabrasion devices, which fall under the Class I category and are exempt from 510(k) requirements.
Some companies manufacturing and selling microneedling devices may register their facilities with the FDA but incorrectly list their devices as Class 1 dermabrasion accessory devices.
As a result, claims that such microneedling devices are “FDA approved” are inaccurate, regardless of the company making the assertion. By understanding the FDA’s classification system for microneedling devices, consumers can make more informed decisions about their aesthetic treatments.
Microneedling Pens and Microneedling: Navigating Classifications and Safety
As the popularity of Microneedling Pen and microneedling treatments continues to grow, it’s essential to understand the nuances surrounding their FDA classification and ongoing research.
Microneedling Pen: A New Technology and Its FDA Classification
Microneedling Pen is a relatively new technology, and as such, it is still undergoing extensive research and clinical trials to assess its efficacy and safety.
Microneedling Pen is considered a Class I medical device, which means it does not require FDA approval to be sold in the United States. While many practitioners confidently use the device and stand by its results, it’s crucial to understand its classification and the safety measures in place.
This situation is not uncommon for emerging treatments, and continued studies will contribute to a more comprehensive understanding of Microneedling Pen’s benefits and potential risks. The ongoing research and evaluation of the device are essential steps in maintaining safety and effectiveness for consumers.
Class I Medical Devices: Microneedling Pen and Counterfeit Products
While Microneedling Pen-branded devices are exclusively available to medical and skin-care professionals, it’s possible to find knock-offs and electric microneedling products from other manufacturers online.
These devices also fall under the Class I medical device category and don’t require FDA approval to be sold in the United States. However, it’s important to be aware of the FDA classification system and the challenges surrounding microneedling devices to make informed decisions and avoid potentially unsafe counterfeit products.
Microneedling Pen Safety
Despite being classified as a Class I medical device, Microneedling Pen has a proven track record of safety and effectiveness when used in professional settings. Proper usage, maintenance, and sterilization of the device are critical in ensuring the safety of the treatment.
It’s worth noting that many other treatments and devices on the market, including some cosmetic procedures, also fall under the Class I category and are widely used due to their effectiveness and minimal risks.
Get Everything You Need for Your Microneedling Pen and More at 20% off Your First Order with Code “20OFF”!
The Microneedling Pen treatment requires only the best equipment to deliver treatments and results your patients will love. Get your best Microneedling Pen equipment and more at FACE Med Store today!
You can create an account here.

Advantages of Using Microneedling Pen
| Benefits | Description |
| Minimally invasive treatment | Microneedling Pen causes minimal damage and discomfort, promoting the skin’s natural healing process. |
| Reduction in scars, wrinkles, stretch marks | Effectively reduces the appearance of scars, wrinkles, and stretch marks by stimulating collagen production. |
| Stimulates collagen production | Encourages the production of collagen and elastin, maintaining skin’s firmness and elasticity. |
| Short recovery time | Requires minimal downtime, allowing patients to resume daily activities soon after treatment. |
| Cost-effective | More affordable compared to treatments such as laser therapy and chemical peels. |
Ensuring the Safe Use of Microneedling Pen: A Detailed Guide
To maximize the safety and effectiveness of Microneedling Pen treatment, it’s crucial to follow these guidelines:
Choose a Reputable Provider
- Research and verify qualifications: Make sure the provider is well-trained, certified, and has experience in performing Microneedling Pen treatments. Check their credentials and look for any relevant certifications or memberships in professional organizations.
- Read reviews and testimonials: Look for reviews and testimonials from previous clients to gauge the provider’s reputation and level of expertise. You can find these on their website, social media profiles, or third-party review websites.
- Schedule a consultation: A face-to-face consultation allows you to discuss your goals and concerns, assess the provider’s communication skills, and determine if you feel comfortable with their approach and expertise.
- Be aware of FDA approval status: While Microneedling Pens fall under the Class I medical device category and don’t require FDA approval, it’s essential to stay informed about the regulatory status of the device and any updates that may arise.
Recognize and Avoid Counterfeit Microneedling Pens
- Be cautious when purchasing Microneedling Pen devices online: Counterfeit Microneedling Pens can be found on various e-commerce platforms. These devices may not be manufactured according to the same quality and safety standards as genuine Microneedling Pens.
- Verify the authenticity of the product: To ensure that you are using a genuine Microneedling Pen, check for the device’s serial number and contact the manufacturer or an authorized distributor to confirm its authenticity.
- Purchase from authorized providers: Make sure to obtain your Microneedling Pen device from a reputable and authorized provider to avoid the risk of using counterfeit products that may compromise the safety and effectiveness of the treatment.
Recognize Potential Side Effects
- Redness and swelling: It’s normal to experience some redness and swelling immediately after the treatment, similar to a mild sunburn. These effects should subside within 24-48 hours.
- Dryness and flaking: The treated area may become dry and flaky as the skin heals. This is a normal part of the healing process and should resolve within a few days.
- Bruising: Although rare, some individuals may experience minor bruising after the treatment. This typically fades within a few days to a week.
- Infection: Infections can occur if the treated area is not properly cared for or if the provider did not follow proper sterilization and usage protocols. Signs of infection include increased redness, pain, pus, or fever. If you suspect an infection, contact your provider immediately for guidance.
Conclusion
Microneedling Pen is a popular and effective skincare treatment classified as a Class I medical device, which doesn’t require FDA approval. To ensure safety and optimal results, it’s important to choose a reputable provider, be aware of FDA regulations, and follow proper guidelines and aftercare instructions. Ongoing research will continue to provide insights into Microneedling Pen’s safety and efficacy, helping individuals make informed decisions about their skincare treatments.
Flawless Skin, Guaranteed Safety: Choose FACE Med Store for Microneedling Pen Products
Are you intrigued by the transformative potential of Microneedling Pen and eager to experience its benefits for yourself? Look no further than FACE Med Store, your trusted partner in skincare and aesthetic treatments.
At FACE Med Store, we understand the importance of safety and efficacy in the world of skincare. That’s why we take pride in offering only the highest quality Microneedling Pen products, meticulously tested to ensure optimal results for our valued clients.
Don’t miss out on the opportunity to reveal your best skin yet. Visit FACE Med Store today and embark on a journey towards radiant, healthy, and rejuvenated skin with our exceptional Microneedling Pen offerings. Let us be your partner in unlocking the true potential of your skin.